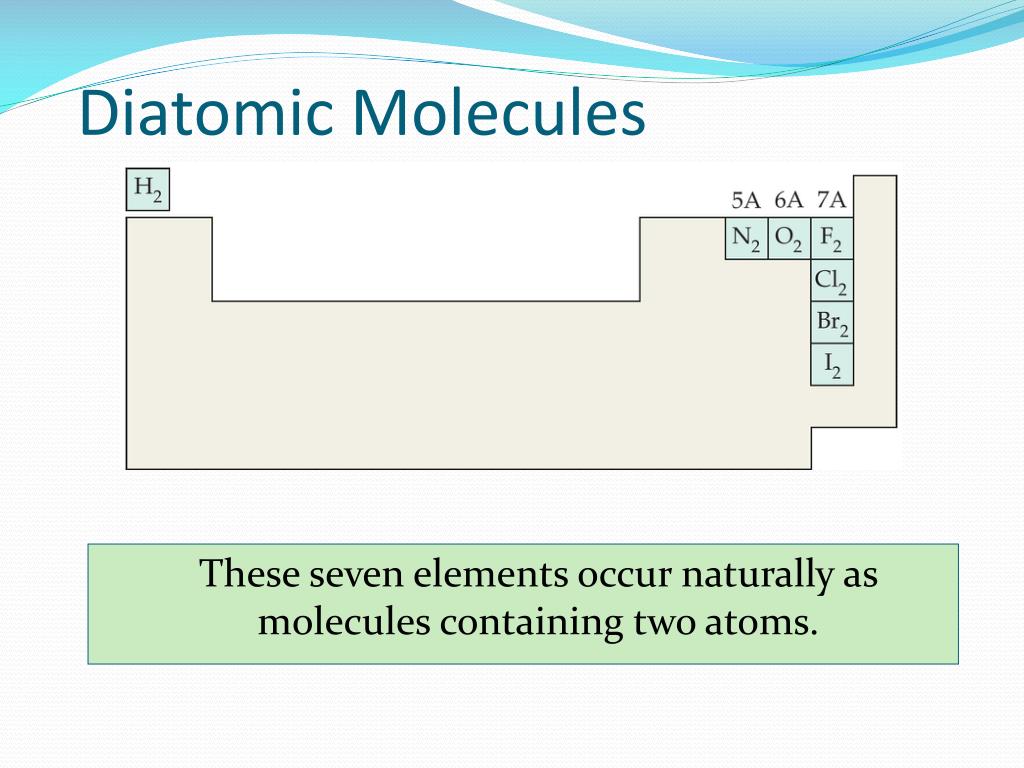
 Let’s say we have a bedroom that looks like this: We just need to pick a room and assign seven distinct spots in it. Step One: Create A Seven Station Memory PalaceĪ Memory Palace is simply a mental recreation of a home, office or other familiar building.Īlthough you can memorize all of these molecules according to location by noticing that everything after hydrogen makes a “seven” shape on the right side of the Periodic Table, we can do better. Note: If you’re really keen, you can use the same technique to memorize the entire Periodic Table of the Elements. 
Let’s look at what it is and how it works step-by-step. You can use it to learn languages much faster, and it will help you never forget a name again.Īnd since you’re dealing with just seven elements, this simple task won’t take long at all. People have used it to memorize and recite entire books backwards and forwards. 
The technique I’m going to teach you has been used for thousands of years. Great! Let’s dig in! How to Remember The Diatomic Elements in 3 Steps Ready to learn the best diatomic elements mnemonic available? You can be a better professional in your career.Īnd you can use the superior memory techniques that get all the information you need into memory for many other aspects of your learning life. How does that acronym help you memorize the name and number of each and every element?Īnd when we do, you can sit for exams with great confidence. Sure, you can use a weak acronym like “Have no fear of ice cold bears,” but You just need a proper mnemonic strategy to get the job done. Figure 4: The ionic radius increases for the generation of negative ions.There are seven diatomic elements and they are very easy to memorize. The gain of an electron adds more electrons to the outermost shell which increases the radius because there are now more electrons further away from the nucleus and there are more electrons to pull towards the nucleus so the pull becomes slightly weaker than of the neutral atom and causes an increase in atomic radius. Figure 3: The ionic radius decreases for the generation of positive ions.Īn anion, on the other hand, will be bigger in size than that of the atom it was made from because of a gain of an electron. A neutral atom X is shown here to have a bond length of 180 pm and then the cation X is smaller with a bond length of 100 pm. This can similarly be said about the protons pulling the electrons closer to the nucleus, which as a result decreases atomic size.įigure 3 below depicts this process. If ten magnets and ten metallic objects represent a neutral atom where the magnets are protons and the metallic objects are electrons, then removing one metallic object, which is like removing an electron, will cause the magnet to pull the metallic objects closer because of a decrease in number of the metallic objects.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
